 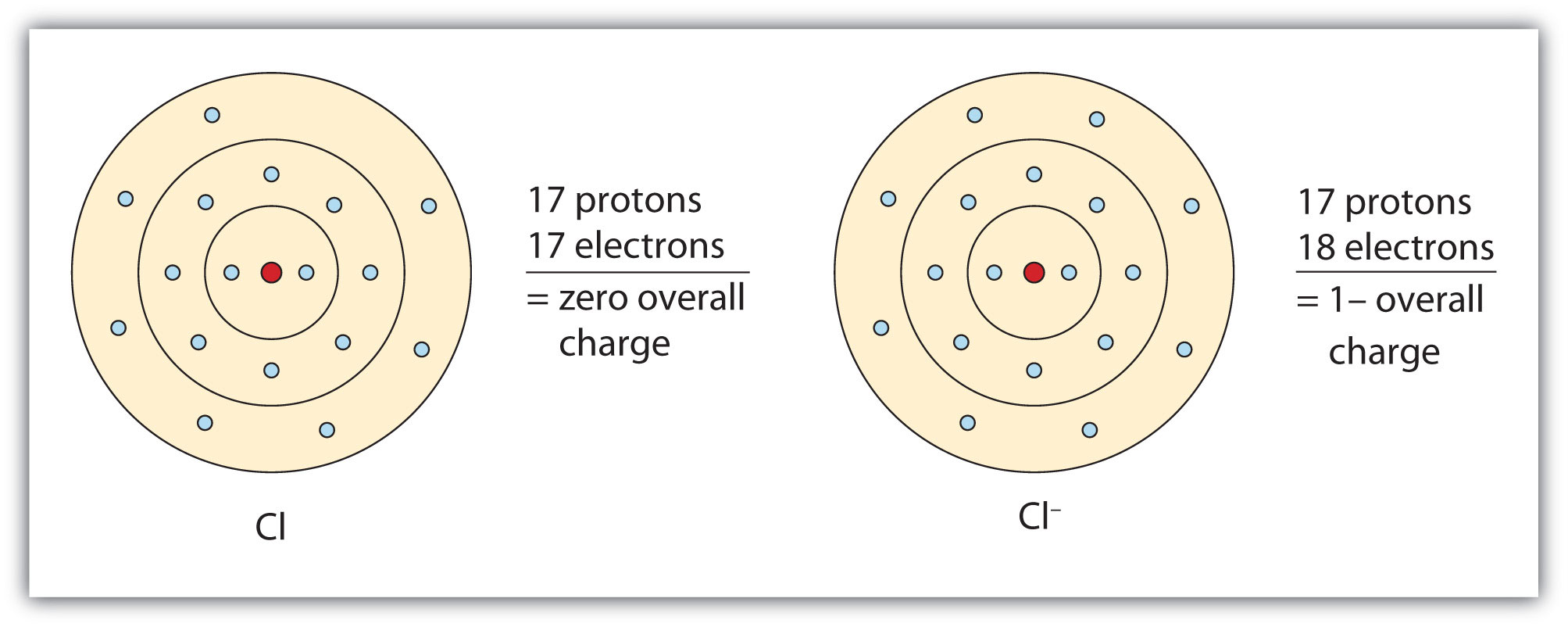
The resulting anion is O 2 − with electron configuration, 1 s 22 s 22 p 6. The second shell has six electrons ( 2 s 22 p 4) and needs two electrons to achieve octet. The electron configuration of O atom is 1 s 22 s 22 p 4. How many electrons must O lose/gain to achieve octet? Write the formula of the resulting ion and its electron configuration. If they got together to form a molecule, there would have to be 2 sodium ions for. 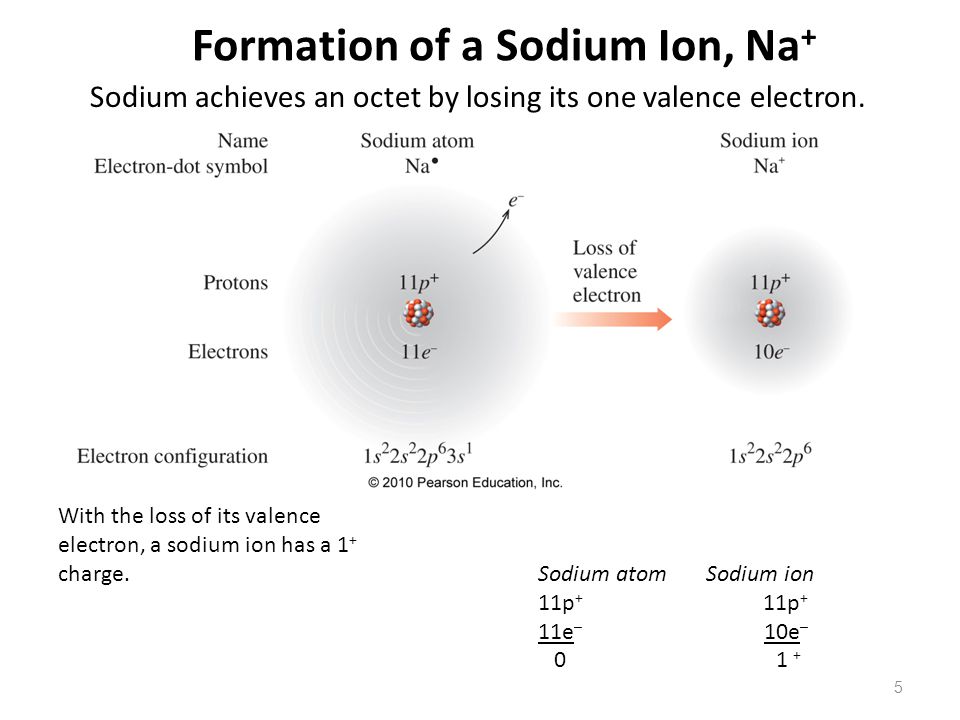
Write the electron configuration of oxygen atom (Z=8). For example Sodium (Na) forms a +1 ion and Sulfur (S) forms a -2 ion. In macroscopic samples of sodium chloride, there are billions and billions of sodium and chloride ions, although there is always the same number of cations and anions. Chloride, by definition, is an anion that has formed from an atom of chlorine. 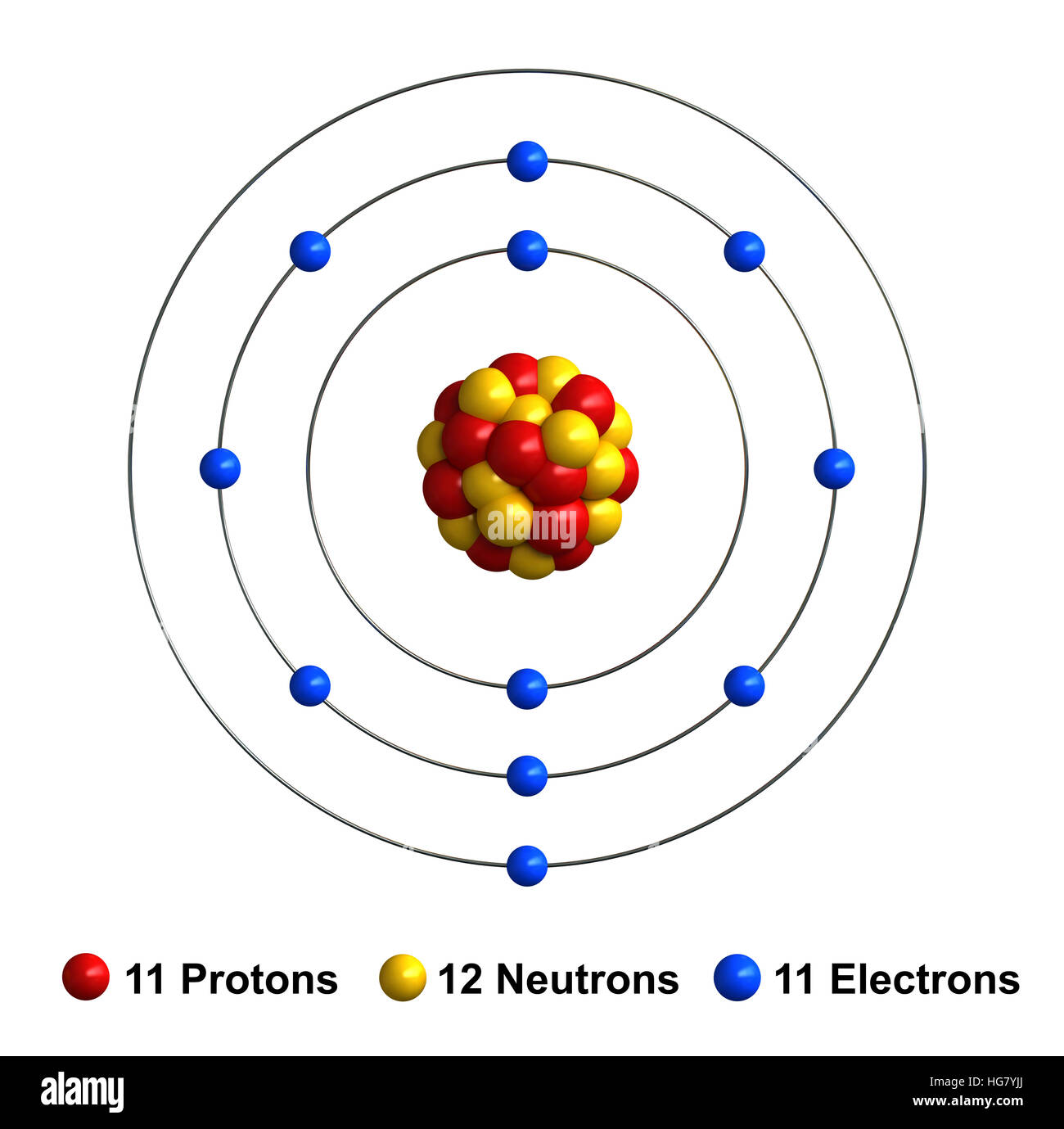
The number of electrons lost by the sodium atom (one) equals the number of electrons gained by the chlorine atom (one), so the compound is electrically neutral. Remember that potassium is a group 1 element that forms a 1+ ion. The pure metal is stored in oil or kerosene because it spontaneously ignites in water. The charge of an electron is considered to be negative by convention and this charge is equal and opposite to the charge of a proton, which is considered to be positive by convention. Notice that there are no leftover electrons. Sodium is a silvery-white metal belonging to Group 1 of the Periodic Table, which is the alkali metals group. An ion ( / a.n, - n /) 1 is an atom or molecule with a net electrical charge. The resulting combination is the compound sodium chloride. These are referred to as the valence orbitals and. Atoms prefer to gain the stability of octet, by having eight electrons in the outer shell, the electrons of the s and p orbitals. In this configuration we note that there is only one electron in the 3rd energy level. For sodium, which is in the 3rd row of the periodic table, the valence electrons will be found in the 3rd shell. The electron configuration of a neutral sodium atom is 1s22s22p63s1. The shell number representing the valence shell will differ depending on the atom in question. With two oppositely charged ions, there is an electrostatic attraction between them because opposite charges attract. Valence electrons are electrons found in the outermost shell of an atom. The names for positive and negative ions are pronounced CAT-eye-ons and ANN-eye-ons, respectively. On the right, the chloride ion has 18 electrons and has a 1− charge. 4 Site name Steamboat Creek Willamette River Basin break - point values Canadian Council of Ministers of the Environment TEL guidelines Sb 2.0 1.3 na As 10. Sodium ion on right has 17 protons and 18 electrons, with a -1 overall charge. The resulting sodium ion has the same electron. On the left, the chlorine atom has 17 electrons. In forming an ionic bond, the sodium atom, which is electropositive, loses its valence electron to chlorine. Within a group (family) of elements, atoms form ions of a certain charge.\): The Formation of a Chlorine Ion. The charge that an atom acquires when it becomes an ion is related to the structure of the periodic table. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
